Coronavirus in Africa: WHO releases updated list of countries with highest cases, deaths
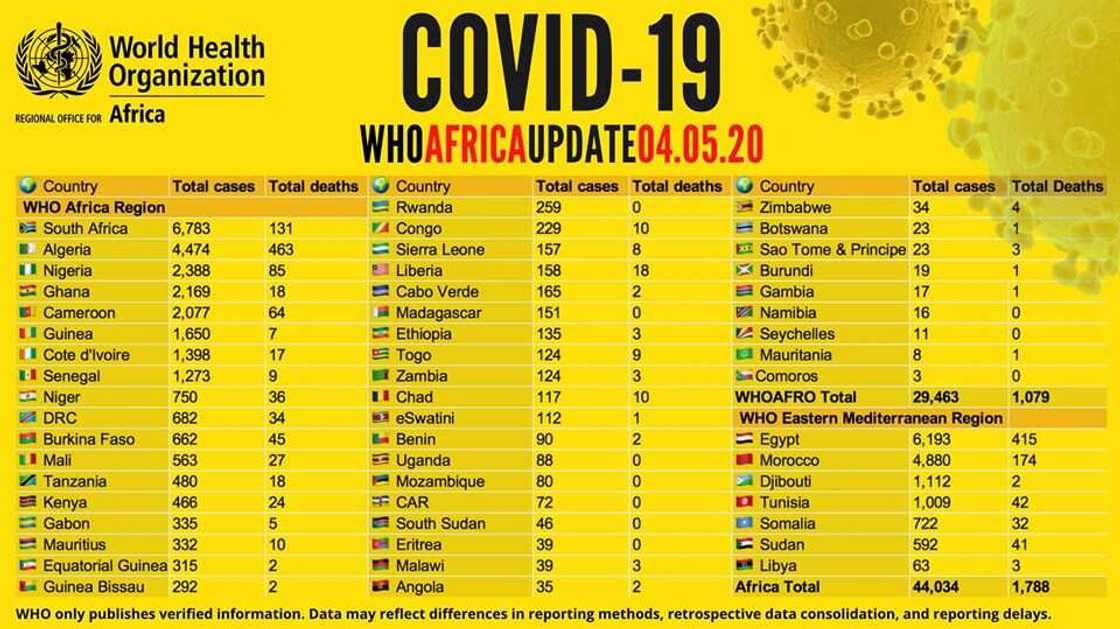
- World Health Organisation's data shows 44,034 confirmed COVID-19 cases have been confirmed on the African continent
- The health organisation also stated that 1,788 COVID-19 deaths have been recorded so far
- According to the data, South Africa has 6,783 cases, the highest coronavirus cases in Africa while Libya has 63 cases, the lowest on the continent
PAY ATTENTION: Click “See First” under the “Following” tab to see Legit.ng News on your Facebook News Feed
More than 44,000 coronavirus cases have been reported in Africa, with over 1,700 deaths, according to the data released by the World Health Organisation (WHO) on Monday, May 4.
According to the international health organisation, South Africa tops the list of African countries with coronavirus cases as 6,783 have tested positive in the country. Out of the over 6,000 cases, 131 people have died.
Algeria occupies the second spot with 4,474 cases and 463 deaths while Nigeria ranks third on the list with 2,388 cases and 85 deaths.
Legit.ng, however, notes that the latest data from the Nigeria Centre for Disease Control (NCDC) indicates that Nigeria now has 2,558 cases and 87 deaths.
See below the list of African countries and their confirmed COVID-19 cases and deaths, according to WHO:
Source: Facebook
PAY ATTENTION: Install our latest app for Android, read the best news on Nigeria’s #1 news app
Meanwhile, Legit.ng previously reported that the United States's Food and Drug Administration (FDA) had authorised the emergency use of the Ebola drug remdesivir for treating the novel coronavirus.
The authorisation means that the anti-viral drug can now be used on people who are hospitalised with severe Covid-19.
A recent clinical trial showed the drug helped shorten the recovery time for people who were seriously ill.
The drug did not, however, significantly improve survival rates. It was originally developed to treat Ebola.
Though it helped reduce the recovery time for COVID-19 patients, experts warned that remdesivir should not be seen as a "magic bullet" for treating coronavirus.
In a meeting with the US President Donald Trump, Daniel O'Day, the chief executive of Gilead pharmaceutical company which developed the drug said the FDA authorisation was an important first step.
He disclosed that the company would donate 1.5 million vials of the drug. According to BBC, emergency FDA authorisation is not the same as formal approval, which requires a higher level of review.
NAIJ.com (naija.ng) -> Legit.ng We have updated to serve you better
Coronavirus: Are Nigerians really afraid of COVID-19? New survey reveals more | Legit TV
Source: Legit.ng



